04 April 2026 | Health & Medicine
The global market for medical bionic implants and artificial organs is set to witness substantial growth in the coming years, driven by rapid technological innovation and rising healthcare demand. Experts attribute the expansion to increasing focus on personalized medical solutions that enhance patient outcomes and quality of life.
Market Forecast and Growth Drivers
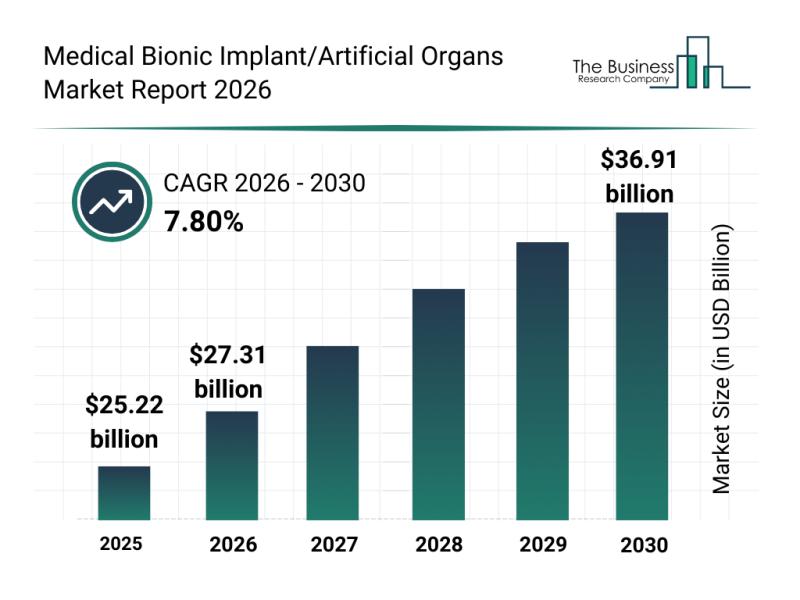
According to industry analysts, the market for medical bionic implants and artificial organs is projected to reach $36.91 billion by 2030, growing at a compound annual growth rate (CAGR) of 7.8%. Key factors driving this growth include:
-
Rising investments in regenerative medicine
-
Increasing use of AI-enabled implant monitoring systems
-
Healthcare needs of an aging population
-
Shift towards minimally invasive implantation techniques
-
Growing demand for smart therapeutic implants
Market trends over the forecast period are expected to include widespread adoption of advanced prosthetic technologies, integration of connected and intelligent implants, expansion of personalized bionic solutions, and a stronger focus on improving long-term implant performance.
Download a free sample of the market report
Leading Companies Driving Market Innovation
Several prominent players are shaping the medical bionic implant and artificial organs sector, including Medtronic PLC, Abbott Laboratories Inc., Cochlear Limited, Ekso Bionics Holdings Inc., Cyberonics Inc., LivaNova PLC, NeuroPace Inc., Ottobock SE & Co. KGaA, SynCardia Systems LLC, Edwards Lifesciences India Pvt. Ltd., and Advanced Bionics AG, among others. These companies are investing in research, product innovation, and advanced implant technologies.
Recent Technological Developments
In April 2024, Science Corporation, a U.S.-based company specializing in brain-computer interfaces and vision restoration, acquired the intellectual property and assets for the PRIMA retinal-implant system from France-based Pixium Vision SA. This acquisition expands Science Corporation’s capabilities in vision restoration and implantable sensory prosthetics, providing access to ongoing clinical trials and advanced implant technology.
Similarly, innovative patient-specific implants are reshaping surgical outcomes. For example, 3D Systems, a U.S. medical device company, received FDA 510(k) clearance in April 2024 for its VSP PEEK Cranial Implant. This 3D-printed cranial reconstruction implant, designed using patient anatomical data, enhances surgical precision, reduces operation times, and improves recovery and functionality.
Market Segmentation
The medical bionic implant and artificial organs market is categorized as follows:
By Type:
-
Bionic Implants: Bionic Limbs, Bionic Eyes, Cochlear Implants
-
Artificial Organs: Artificial Hearts, Artificial Kidneys, Artificial Livers, Artificial Pancreas
By Technology: Electronic, Mechanical
By Application: Hospitals, Clinics
These segments highlight the diverse applications and technological advancements that continue to propel market growth.